HS271
CAS No. 2410393-15-4
HS271 ( —— )
产品货号. M28114 CAS No. 2410393-15-4
HS271 是一种高效、口服活性的选择性 IRAK4 抑制剂,IC50 为 7.2 μM。 HS271 表现出卓越的酶活性和细胞活性,以及优异的药代动力学特性。
纯度: >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| 规格 | 价格/人民币 | 库存 | 数量 |
| 5MG | ¥2341 | 有现货 |


|
| 10MG | ¥3872 | 有现货 |


|
| 25MG | ¥6391 | 有现货 |


|
| 50MG | ¥8748 | 有现货 |


|
| 100MG | ¥11988 | 有现货 |


|
| 500MG | ¥24057 | 有现货 |


|
| 1G | 获取报价 | 有现货 |


|
生物学信息
-
产品名称HS271
-
注意事项本公司产品仅用于科研实验,不得用于人体或动物的临床与诊断
-
产品简述HS271 是一种高效、口服活性的选择性 IRAK4 抑制剂,IC50 为 7.2 μM。 HS271 表现出卓越的酶活性和细胞活性,以及优异的药代动力学特性。
-
产品描述HS271 is a highly potent, orally active and selective IRAK4 inhibitor, with an IC50 of 7.2 μM. HS271 exhibits superior enzymatic and cellular activities, as well as excellent pharmacokinetic properties.(In Vivo):In rat models of LPS-induced TNFα production collagen-induced arthritis, HS271 (15-150 mg/kg) shows robust antiinflammatory efficacy with a t1/2 of 3.3 h and Cmax of 2107 ng/mL. The oral bioavailability of HS271 is 67.3%, 58.2%, 14.4&% and 49% in mice, rats, dogs, and monkeys, respectively.
-
体外实验——
-
体内实验HS271 (15-150 mg/kg) displays robust in vivo antiinflammatory efficacy as evaluated in rat models of LPS induced TNFα production collageninduced arthritis.HS271 exhibits a t1/2 of 3.3 h and Cmax of 2107 ng/mL.HS271 is stable in liver microsome assays across other species, including rat, mouse, monkey, and human.HS271 exhibits oral bioavailability of 67.3%, 58.2%, 14.4&% and 49% in mouse, rat, dog and monkey, respectively. Animal Model:A rat model of collagen induced arthritis (CIA).Dosage:15, 50, 150 mg/kg.Administration:PO, once daily.Result:Led to a significant reduction in paw swelling as compared to vehicle control, with a minimum effective dose at 15 mg/kg QD. Notably, at 150 mg/kg QD, HS271 eliminated the paw swelling.
-
同义词——
-
通路Others
-
靶点Other Targets
-
受体G2019S LRRK2|LRRK2
-
研究领域——
-
适应症——
化学信息
-
CAS Number2410393-15-4
-
分子量435.44
-
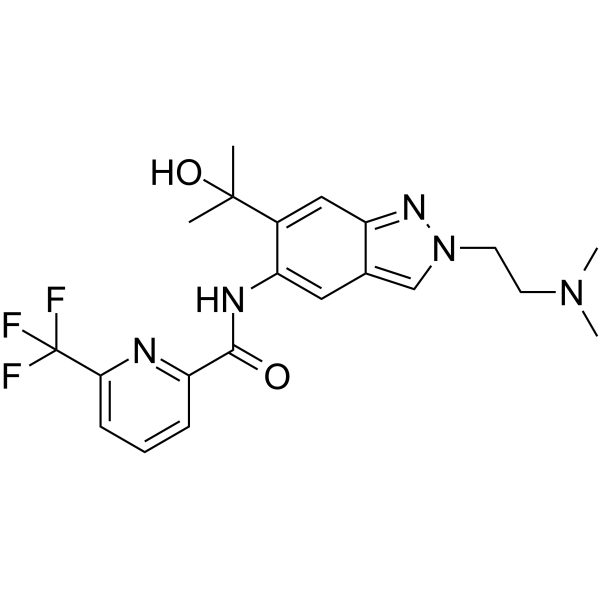
分子式C21H24F3N5O2
-
纯度>98% (HPLC)
-
溶解度In Vitro:?DMSO : 100 mg/mL (229.65 mM)
-
SMILESN/A
-
化学全称——
运输与储存
-
储存条件(-20℃)
-
运输条件With Ice Pack
-
稳定性≥ 2 years
参考文献
1.Nigel Ramsden, et al. Chemoproteomics-based design of potent LRRK2-selective lead compounds that attenuate Parkinson's disease-related toxicity in human neurons. ACS Chem Biol. 2011 Oct 21;6(10):1021-8.
产品手册




关联产品
-
Proinsulin C-peptide...
Proinsulin C-peptide (human) is a 31-amino acid peptide that connects the Proinsulin a and B chains, ensuring its proper folding, biological activity, and regulation of cellular functions.
-
Itepekimab
Itepekimab (REGN-3500) 是一种 IL-33 的单克隆抗体。Itepekimab 在前期临床研究中可减少气道炎症和相关组织损伤。Itepekimab 在哮喘、慢性阻塞性肺病 (COPD) 和特应性皮炎 (AD) 中具有潜在作用。
-
β-Endorphin (1-27), ...
β-Endorphin (1-27) (human) 是一种 opioid 拮抗剂,结合 μ-、δ- 和 κ-阿片受体,其 Kis 分别为 5.31、6.17 和 39.82 nM。β-Endorphin (1-27) (human) 抑制 β-Endorphin 诱导和 etorphine 诱导的痛觉缺失。



 021-51111890
021-51111890 购物车()
购物车()
 sales@molnova.cn
sales@molnova.cn







